THE ELECTRON MICROSCOPE: A WINDOW INTO THE NANOSCOPIC UNIVERSE
By: Laura Ruhge
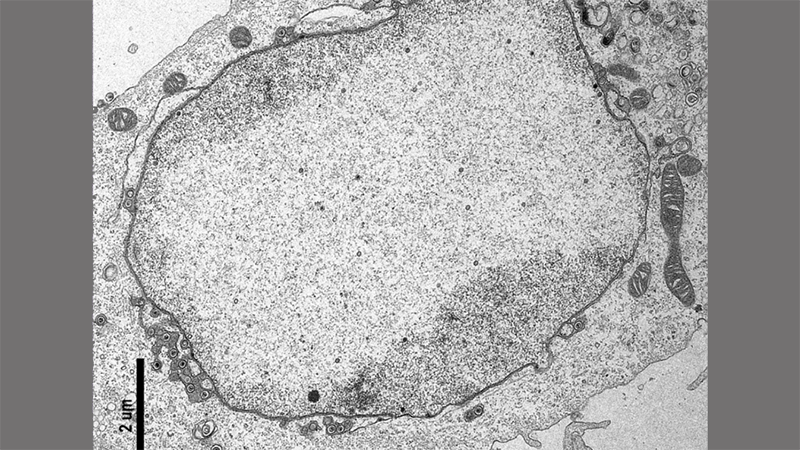
Cell infected with herpes simplex virus type 1. Image captured by the author in the Center for Advanced Microscopy core facility at Northwestern University Feinberg School of Medicine. The black scale bar is 2 micrometers (um) long which is 250 times smaller than a grain of salt. (Photo credit: Laura Ruhge).
I sit in the pitch-black room, twisting the knobs on the control pad of the electron microscope. I’m using this device to hunt for cells. Looking through the eyepiece, I work to bring the shadowy mass into focus. Various shapes and lines within the mass become bolder, more defined. I stare intensely at the heavy, curving lines of the cell’s membranes, my satisfaction increasing as each click of the knob makes them clearer.
Working on this microscope is bewitching. The powerful device towers high on its desk, altogether standing as tall as the room’s doorframe. I gaze through the round viewing screen at its base. Immersed in darkness, my every sense becomes wholly devoted toward driving this strange spaceship-like machine.
Staring through the viewing window, I use the trackball on the control pad to steer my field of vision through the specimen. I pass entire cells like they are a multitude of stars and planets comprising this biological universe. I’m in search of a particular view of a virus-infected planet.
On this kind of microscope, a transmission electron microscope, a viewable sample is cut into ultra-thin slices so different views of a cell are visible. It’s like if you cut a loaf of raisin bread horizontally instead of vertically: depending on how far down the loaf you slice, you get different snapshots of what’s inside. In a cell sample, you may get the very top of a cell, or a slice right through the middle of the cell. I’m searching for virus-infected cells sliced through the middle exposing their insides and, most importantly, a wasteland with hundreds of virus particles littered within.
The hunt for this viral wasteland was not possible until the mid-1900s. Then, light microscopes served as a researcher’s window into the unseen. Human eyes examined the building blocks of life, such as cells nearly 20 times smaller than a grain of salt.
But light microscopy had its limitations. The resolving power, or ability to distinguish separate objects close together, is restricted by the (relatively) long wavelengths of light. Imagine looking at a night’s sky and you observe a single bright star. Then you use a telescope for a closer look and realize what you thought was a single star is actually two stars very close together. Working with a light microscope is like looking at the night sky with your naked eye: to get better resolution, we needed more powerful instruments. Scientists were provided a taste of the microscopic world, but they craved to see more. They could observe a cell under a light microscope, but the contents within that cell were still an unresolved mystery. Out of this desire, the electron microscope was born
The electron microscope, as you may guess by its name, uses electrons to visualize a sample. Electrons are the definition of small – they are the negatively charged particles that, like the moons of a planet, orbit the around the center of the most basic unit of matter, the atom. Because of their negative charge, electrons are manipulated by a magnetic field. Remember how that magnifying glass you held as a kid captured the sunlight into a focused, bright spot (perhaps you were burning ants)? The same concept can focus a beam of electrons.
In 1926, Physicist Hans Busch theoretically demonstrated a short coil generating a magnetic field would result in electrons behaving like light does when passed through a converging lens: the electrons could be focused into a beam. Busch suggested that magnetic fields like this would be able to direct a beam of electrons, perhaps to obtain images, but he never tested his theoretical optics.
A few years later, a young and eager engineering student named Ernst Ruska read Busch’s paper. Building an electromagnetic lens to magnify an object was a worthy challenge. In 1931, Ruska tested this theory and successfully magnified an image of a specimen by blasting it with a beam of electrons focused through an electromagnetic lens.
Several years later, Ruska built the first transmission electron microscope. It wasn’t perfect, but Ruska’s microscope made sense of detail 10,000-times smaller than a grain of salt. The electron beam’s shorter wavelength enabled Ruska’s device to resolve details 4-times smaller than even the best modern light microscope can achieve even today.
Fast forward to the present. Today, electron microscopes are capable of magnifying an object up to 2-million times, making sense of details smaller than a nanometer. That’s 500,000-times smaller than a grain of salt. Dr. Ruska’s invention enabled researchers to see viruses, DNA, and eventually even atoms – all of which were invisible to light microscopes.
There it is. I’ve found my mark. I click the magnification knob of the control pad and in my vision field, the planet cell gets closer. Some more finessing brings it into focus. There. I now see this cell is infected. Virus particles 4,000-times smaller than a grain of salt are scattered about inside, their hexagonal shells crisp and clear. I’ve found the perfect view of my viral wasteland. Thank you, Dr. Ruska, your invention opened our eyes to the nanoscopic universe within.
